Tacrolimus is a potent immunosuppressive macrolide medication primarily used to prevent organ rejection in transplant recipients. It works by inhibiting T-lymphocyte activation. Available in oral and intravenous formulations, it requires careful therapeutic drug monitoring due to its narrow therapeutic index and potential side effects, such as nephrotoxicity and neurotoxicity.

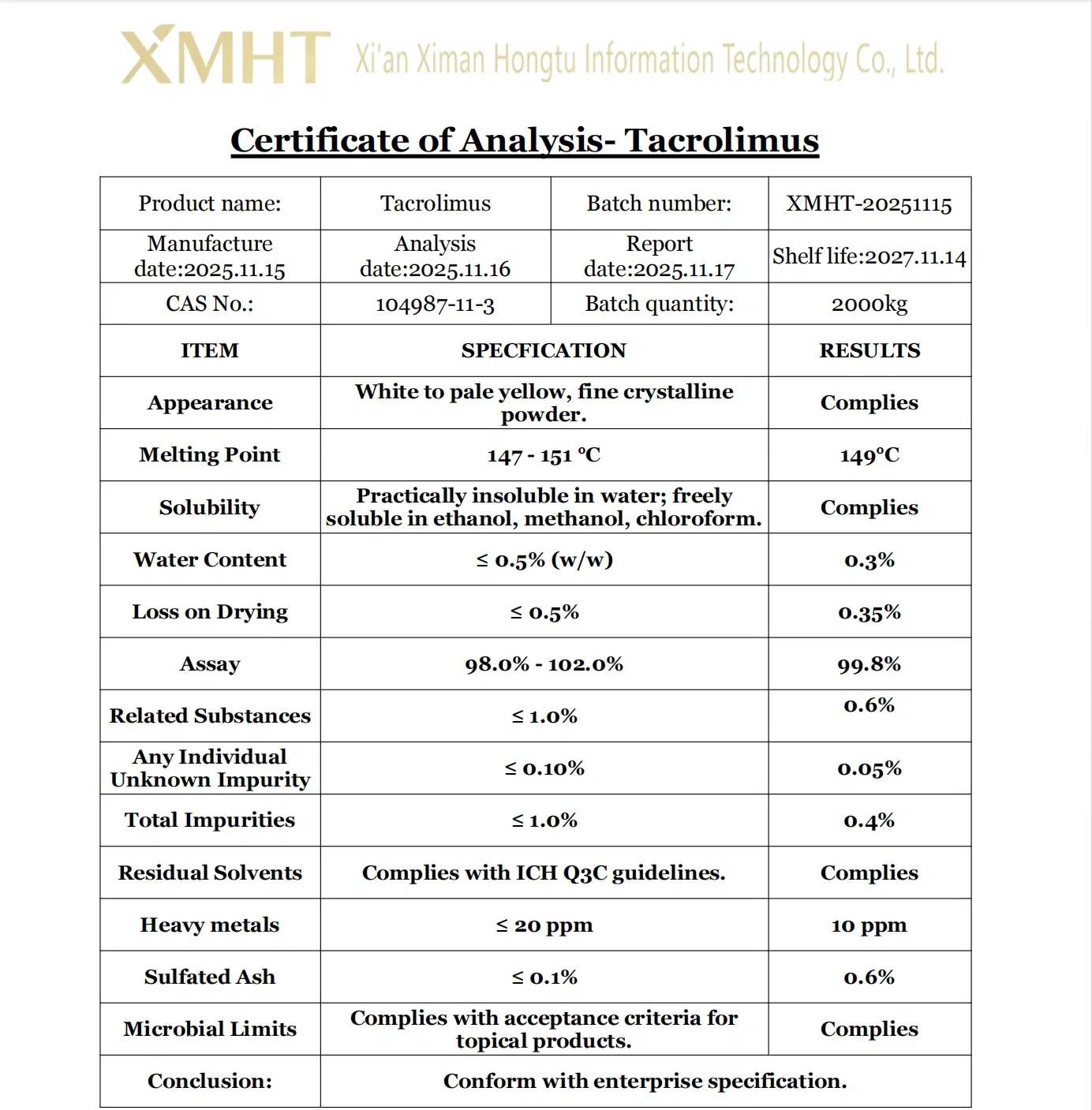
| Product Name | Tacrolimus |
| Appearance | White Powder |
| CAS | 104987-11-3 |
| MF | C44H69NO12 |
| MW | 804.031 |
Tacrolimus exerts its effect by selectively inhibiting calcineurin, a critical phosphatase enzyme within T-lymphocytes. This prevents the dephosphorylation of NFAT, thereby blocking the transcription of genes essential for T-cell activation, such as interleukin-2 (IL-2). By suppressing IL-2 production, Tacrolimus effectively dampens the cellular immune response.
Beyond transplantation, topical Tacrolimus is a pivotal, steroid-sparing agent in dermatology for managing atopic dermatitis (eczema). It provides a therapeutic option for sensitive areas like the face and neck, reducing pro-inflammatory cytokines that cause pruritus, erythema, and lichenification without the risks associated with long-term corticosteroid use.